
“The thermodynamic punishment must fit the structural crime.”
Research Vision:
Humankind has an innate appetite for exploration, energy, and speed. These areas all require materials that operate in the most extreme environments, such as high temperatures (>1500 ˚C). While exploring the universe can be cold (~-270 ˚C in deep space), re-entry into a planet’s atmosphere can be hot (~1500 ˚C on Earth). Thus, high temperature thermal barrier systems are required. Energy production through nuclear fission can reach temperatures of up to ~1700 ˚C and even higher for nuclear fussion, while nuclear thermal propulsion systems require temperatures up to ~2800 ˚C to provide thrust to propel next generation spacecrafts to Mars and beyond. When traveling at hypersonic speeds on Earth, leading edges can reach temperatures of ~2700 ˚C (at Mach 8). These are just some of the examples of where next generation high temperature material systems are required to propel civilization into the future.
To effectively design high temperature material systems, one must have a clear understanding of their thermochemical and thermophysical properties, in addition to their atomic structure! The McCormack Laboratories central vision is to investigate and document material systems in extreme environments that enable next generation exploration, energy and high speed technologies
Research Scope:
To develop and document actinide, oxide, carbide, nitride and di-boride thermochemical and thermophysical properties under extreme conditions for next-generation space exploration materials. This leads to 4 key integrated fundamental research themes: (i) Material Synthesis, (ii) Crystallography, (iii) Calorimetry and (iv) CALPHAD. These themes are related to 2 key research initiatives: (i) Fundamental and (ii) Applied. These initiatives will work to support the key objective of materials to support space exploration, hypersonic platforms and nuclear fission/fusion reactors.

Fundamental
The core scientific questions that are of interest to the McCormack laboratory are centered around atomic bonding in extreme environments. Specifically, what is the nature of bonding at the highest of temperatures, on the precipitous of melting and beyond. Core scientific questions of interest for actinides, carbides, nitrides and di-borides include:
(i) What is the local structure prior to melting and after melting?
(ii) What is the atomic mobility prior to melting?
(iii) What is the strength of the atomic bonds in the highest melting point materials?
(iv) How do the 5f electrons in actinides influence bonding, and how are they different compared to surrogate transition metals?
(v) How can the solid and the melt be tailored to increase the melting point beyond the highest known: (Hf.8Ta.2)C (Tmelt. = ~3942 ̊C) and highest predicted: Hf.53C.27N.20 (Tmelt. = ~4142 ̊C)
Applied
The core engineering problems that are of interest to the McCormack laboratory are centered around designing systems to study and understand materials in extreme environments. Specifically, designing, building and maintaining unique, high temperature equipment, calorimeters and materials databases. Core engineering questions of interest include:
(i) To develop novel high temperature experimental systems that can be incorporated with materials diagnostics e.g. in-situ X-ray diffraction, µ-CT, etc…
(ii) To improve temperature measurements at the highest of temperatures
(iii) To develop phase diagrams
(iv) To measure thermochemical (enthalpy of formation, enthalpy of mixing, heat capacity and melting points) and thermophysical properties (molar volume, thermal expansion) at the highest of temperature for engineering design.
(v) To support CALPHAD databases for high temperature materials (actinides, carbides, nitrides and di-borides)
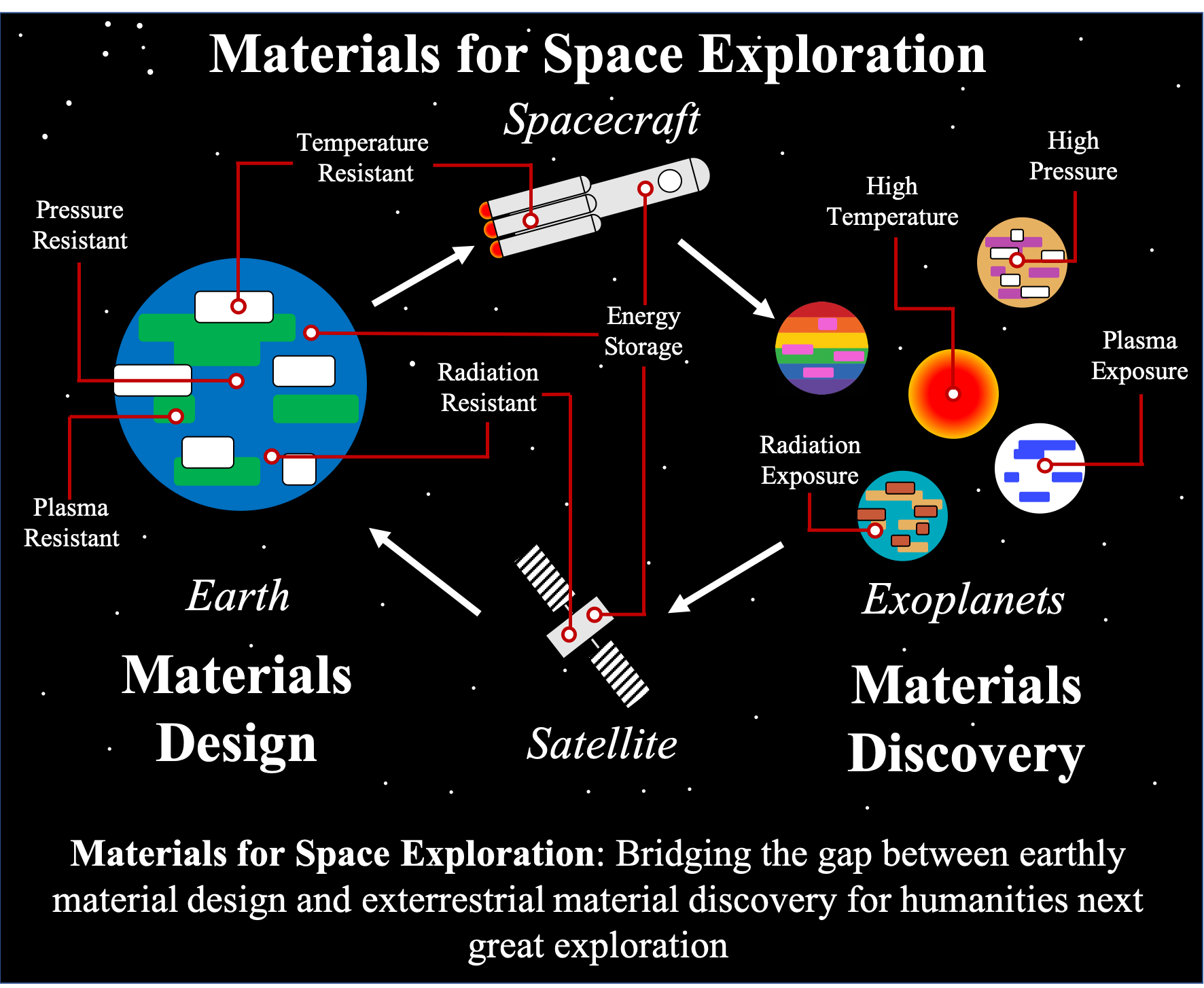
Research Themes:
New complex oxides, carbides, and nitrides will be synthesized using the steric entrapment method. The anisotropic thermophysical properties, viz., thermal expansion, compressibility and molar volume strains (related to the partial molar volume of mixing) will be measured using in-situ powder diffraction as a function of environment e.g. temperature, pressure etc. These thermophysical parameters will be documented for engineering applications and will be used to determine crystallographic orientation relations and lattice variant deformations (related to changes in shape and volume) of any observed transformations. This will be complemented with symmetry decomposition analyses to completely describe the symmetry relations between crystal structures. The phase transformation data will be used to assess if these materials can potentially be used as transformation toughening or shape memory ceramics. Thermochemical properties, viz., heat capacity, enthalpy of formation, the entropy of formation and enthalpy of mixing will be measured to determine the stability of the newly synthesized compounds. This information will influence the choice of future compositions for potential applications as high temperature, energy storage or structural materials. Finally, with the aid of the CALPHAD method, the complex oxide, carbide and nitride phase diagrams will be produced and optimized based on the experimentally measured thermophysical and thermochemical data.
This work is a multidisciplinary endeavor, aimed at merging ideas from: (i) materials synthesis, (ii) crystallography (advanced symmetry and experimental analysis), (iii) calorimetry and (iv) CALPHAD to help tackle the space exploration challenge through the identification and fundamental understanding of new extreme environment for applications in energy storage, structural and shielding materials.

Articles that inspire our work
1. Materials Research for Fusion (2016)
2. Space Nuclear Propulsion for Human Mars Exploration (2021)
3. US hypersonic initiatives require accelerated efforts of the materials research community (2021)
Lab Values:
Innovation | Diversity | Comradery | Humility | Self-Improvement